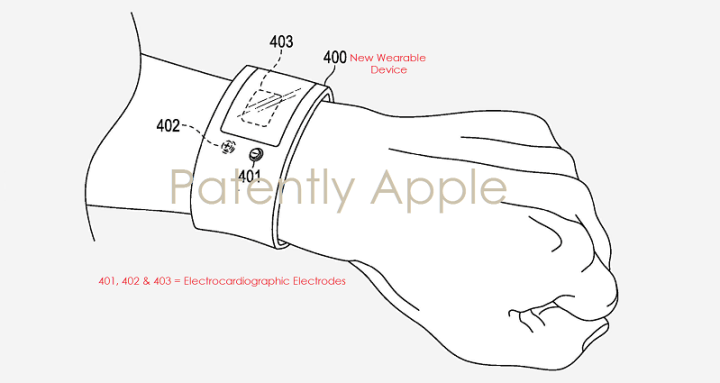
In fact, according to the latest reports from Patently Apple, the company’s new wearable may be a heart reader, pushing the company further into the medical field than it already is. A patent filed by Apple shows that the new wearable would be worn on the wrist or another part of the body, and used to track everything about the heart.
For example, the device would be able to perform an electrocardiogram to measure the heart’s rhythm, and would be targeted more at those with heart problems than the average consumer.
The Apple Watch is no slouch itself when it comes to monitoring vitals. Thanks to a bevy of sensors including a heart rate sensor and pedometer, it can paint a fairly holistic picture of a wearer’s physiology. But Apple’s next wearable might offer biological tracking of a far more granular sort. Economic Daily News reports that it’ll record variations in glucose and blood oxygen level, in addition to heartbeat rate and pulse.
The secretive effort is apparently being spearheaded by Apple’s Jay Blahnik, Apple’s director of fitness and health technologies who, prior to joining Apple, was heavily involved in the development of Nike’s Nike+ Running, FuelBand, and Kinect Training fitness platforms. Research and development of the wearable has reportedly been underway for “more than two years.”
Rumblings of a revolutionary new health tracker line up with Apple’s recent hiring spree. In early June, the company recruited Dr. Rajiv Kumar, a pediatric specialist and endocrinologist at Stanford Children’s Hospital, to work on unspecified “health projects.” Kumar made headlines in 2014 when he used Apple’s ResearchKit platforms in tandem with an iPod touch to monitor blood sugar levels in Type 1 diabetes patient.
And in May, Apple posted a job listing for a Privacy Counsel whose responsibilities would include, among other things, ensuring that the company remained in compliance with the Health Insurance Portability and Accountability Act (HIPPA) — the United States law that provides privacy and provisions for safeguarding medical information.
The Apple Watch isn’t regulated as a medical device by the Food and Drug Administration (FDA), but the company’s upcoming device might be. Apple CEO Tim Cook told The Telegraph last year that it would work with officials on certification “in the future,” comments which followed a Wall Street Journal report detailing the company’s scrapped vision of a futuristic health monitor. Reportedly, Apple engineers planned to pack the first-gen Apple Watch with skin conductivity sensors and EKG monitors, but couldn’t nail down a configuration that could contend with “hairy arms,” “dry skin,” and other conditions.
Whatever the nature of the Cupertino company’s new smart wearable, chances are it’ll take a radically different form than the Apple Watch’s rumored follow-up. In a brief to investors earlier this year, Apple analyst Ming-Chi Kuo said the so-called Apple Watch 2 would feature a “streamlined” design, slightly faster processor, and improved water resistance. The report mentioned sensors as a possibility, too — most likely a GPS and barometer, apparently. And the new Apple Watch might trade the current-generation model’s OLED display technology for an LCD panel of the same size and thickness.
Updated on 08-12-2016 by Christian de Looper: Added information about leaked patent.
Editors' Recommendations
- Here’s how Apple could change your iPhone forever
- One feature will make the Apple smart ring worth waiting for
- An Apple smart ring could be coming soon
- Please stop wearing your Apple Vision Pro outside
- Apple is updating one of the oldest apps on your iPhone


