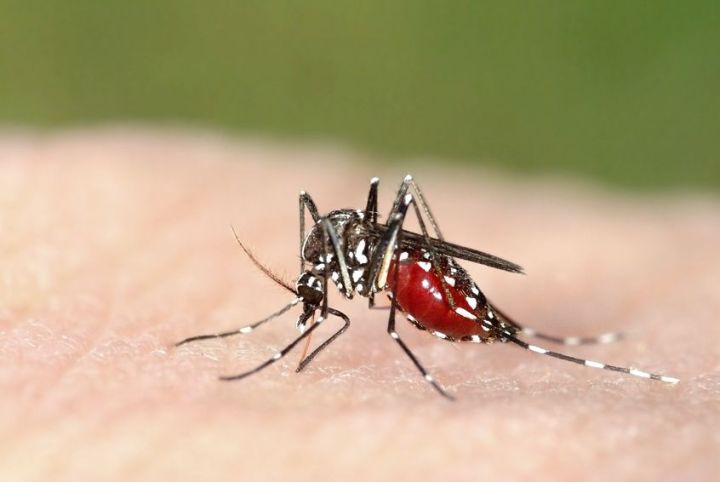
Aedes aegypti is the specific species of mosquito responsible for carrying and spreading the Zika virus. Although the virus usually only manifests as a mild rash on the skin so far, Zika has been linked to 4,000 cases of Brazilian children born with microcephaly, a birth defect that causes infants to be born with abnormally small heads. The same type of mosquito has been known to transmit chikinguya and dengue viruses – dengue alone has led to illnesses in 100 million people every year.
The gene drive is very new technology, and if administered to the population of aedes aegypti mosquitos, it would work more quickly and effectively to existing solutions. For example, biotech company Oxitec has created a genetic mutation that would kill off the mosquito population by sterilizing males and creating offspring that die before reaching reproductive age. This method is less effective than the gene drive technique, because it relies on the organic process of gene selection through reproduction. Alternatively, gene drive solution would spread the lab-designed changes to every single mosquito descendent of the engineered breed instead of only some. Then, the gene drive would either eradicate the species entirely or make it unable to spread the virus depending on the specific changes to the mosquito’s genetics.
Three labs in the United States are already working toward a gene drive to alter the Aedes aegypti species, but ecologists are starting to push back on the concept. With an eye on the world’s ecosystem overall, eradicating an entire species that functions as a part of the whole is a dangerous prospect for ecologists. It’s possible that the sterilizing “population replacement” approach could calm these concerns, instead of the more drastic species eradication method. In population replacement, the gene drive would spread a genetic change that would make the aedes aegypti mosquitos unable to host the pathogen that is infecting humans.
University of California, Irvine hosts one of the labs working on a gene drive to stop Zika, and they believe they could have the technology perfected within a year. Unfortunately, the consequences of messing with genetics are posing enough concerns that getting the necessary political and governmental approvals to use the gene drive in the real world could take considerably longer.


