Nothing lasts forever — especially not smartphone batteries. Those tiny lithium-ion powerhouses that run your iPhone or Android phone are all consumables, which means they have a limited life span. The longer you hang on to your device, the more your battery will degrade, and eventually, it will have to be replaced.
Smartphone batteries don’t usually just conk out. There are telltale hints over time, mostly relating to your phone’s performance. You may notice that the device is slow to respond to taps and prompts or that apps take longer to launch or respond to commands. Or, perhaps your phone drains more rapidly than in the past, is slower to power up, never powers up to 100%, shuts down unexpectedly under normal conditions, or will not run unless it is plugged into the charger. Other more ominous signs include the phone heating up while charging or feeling thicker (perhaps the result of a ruptured battery), or perhaps there are no signs of life no matter what you do. Any of these circumstances can signal a failing, dead, or even hazardous battery.
Current iPhone and Android operating systems can provide some hints to warn you of the demise of your battery that may prepare you for the ultimate decision about what to do. Sadly, changing
Apple provides a host of tools in its more recent operating systems that assist you in determining the health of your battery and give you solid intelligence on whether or not it needs replacing anytime soon. For
How to tell if your iPhone battery needs replacing
In late 2017, Apple acknowledged it had deliberately and secretly throttled some of its older phones to stabilize performance caused by aging batteries. Starting with iPhone 6 and iOS 11.3 you can go to Settings > Battery > Battery Health to find a tool that reveals significant information about the current condition of your battery, and recommends if a battery should be replaced. You can also disable the performance management feature, though you may risk unexpected shutdowns if your battery is degraded.
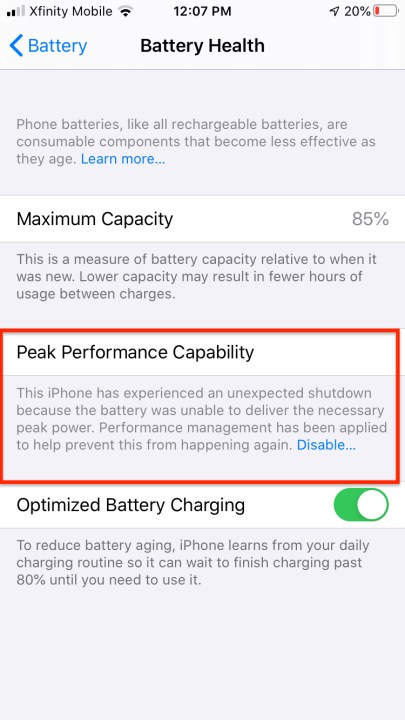
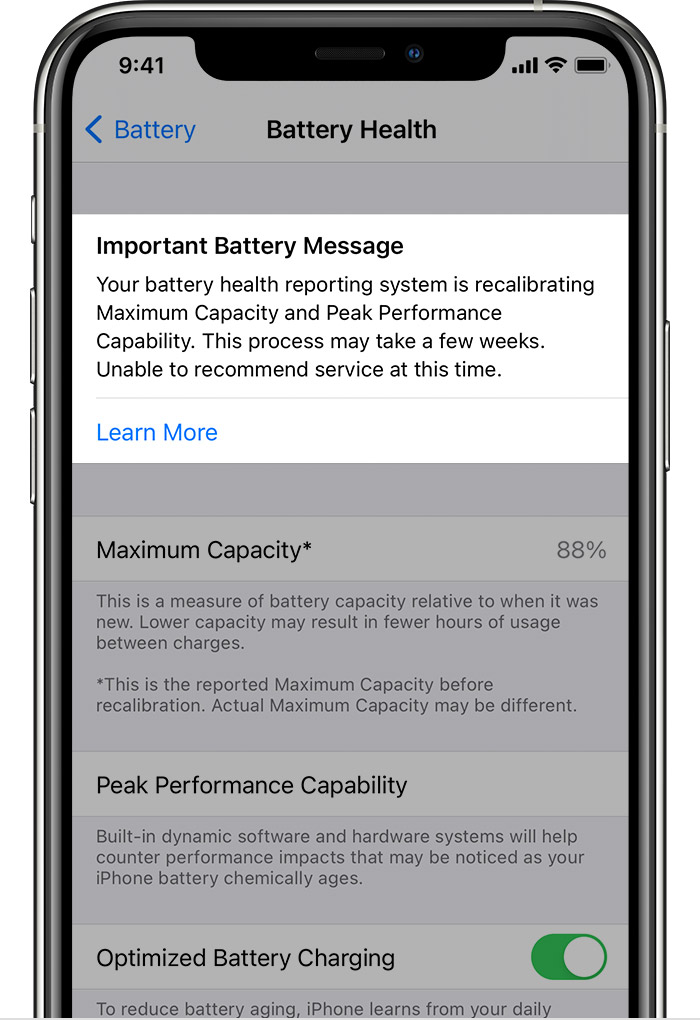
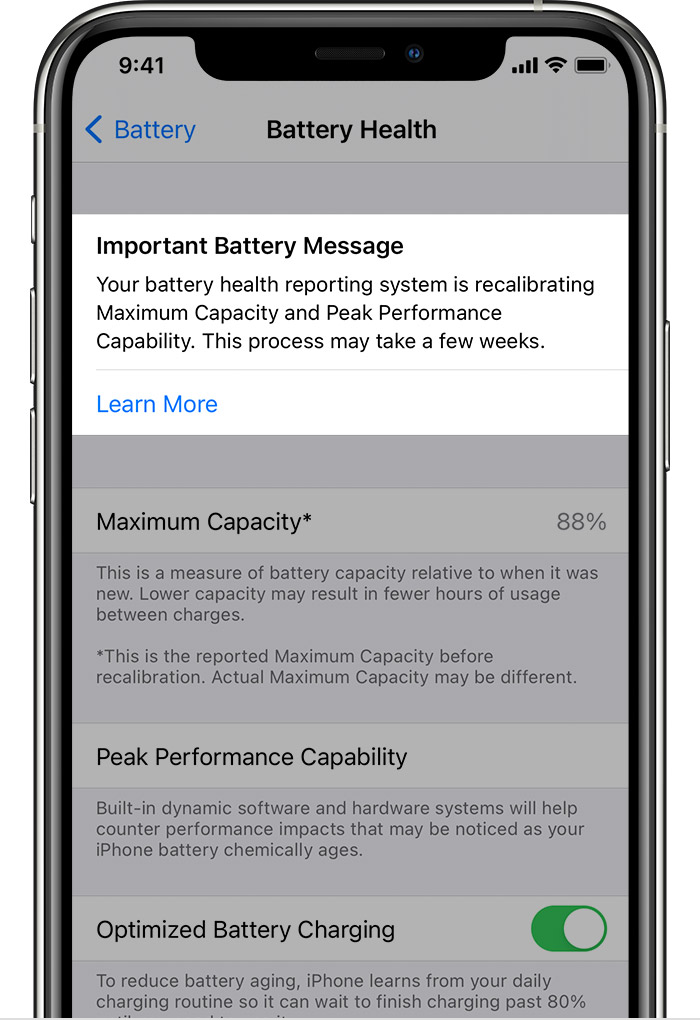
When you tap on Battery Health, you can easily see the relevant statistics. Maximum Capacity is a percentage that measures current capacity versus 100% when your battery was new. A range of that percentage is normal, though a healthy battery should retain about 80% of its original capacity at 500 charge cycles. If capacity falls below 80%, Apple recommends replacing the battery.
You can observe whether the performance management feature, which manages the iPhone’s maximum performance and prevents shutdowns, is enabled and you can switch it off on any iPhone 6, 6 Plus, 6s, 6s Plus, SE, 7, and 7 Plus. This feature is enabled only after an unexpected shutdown on a device whose battery peak power is diminished. If you disable performance management, you can’t turn it back on, but it will automatically start up again in the event of another unexpected shutdown. The feature is included with iPhone 8, 8 Plus, and X with iOS 12.1, and in
The iPhone’s Battery Health feature can deliver several messages that give you a good idea of the current condition of your battery. Below are the messages verbatim. Message number four is the only one that specifies that your battery is severely degraded and should be replaced.
- Your battery is currently supporting normal peak performance.
- This iPhone has experienced an unexpected shutdown because the battery was unable to deliver the necessary peak power. Performance management has been applied to help prevent this from happening again. Disable…
- This iPhone has experienced an unexpected shutdown because the battery was unable to deliver the necessary peak power. You have manually disabled performance management protections.
- Your battery’s health is significantly degraded. An Apple Authorized Service Provider can replace the battery to restore full performance and capacity. More about service options…
- Unable to verify this iPhone has a genuine Apple battery. Health information not available for this battery. Learn more…
- This iPhone is unable to determine battery health. An Apple Authorized Service Provider can service the battery. More about service options…
Replacing your iPhone battery is not cheap: It costs $49 for older iPhones or $69 for the
Recalibration of battery health report in iOS 14.5
There are some special updates for users of iPhone 11, iPhone 11 Pro, and
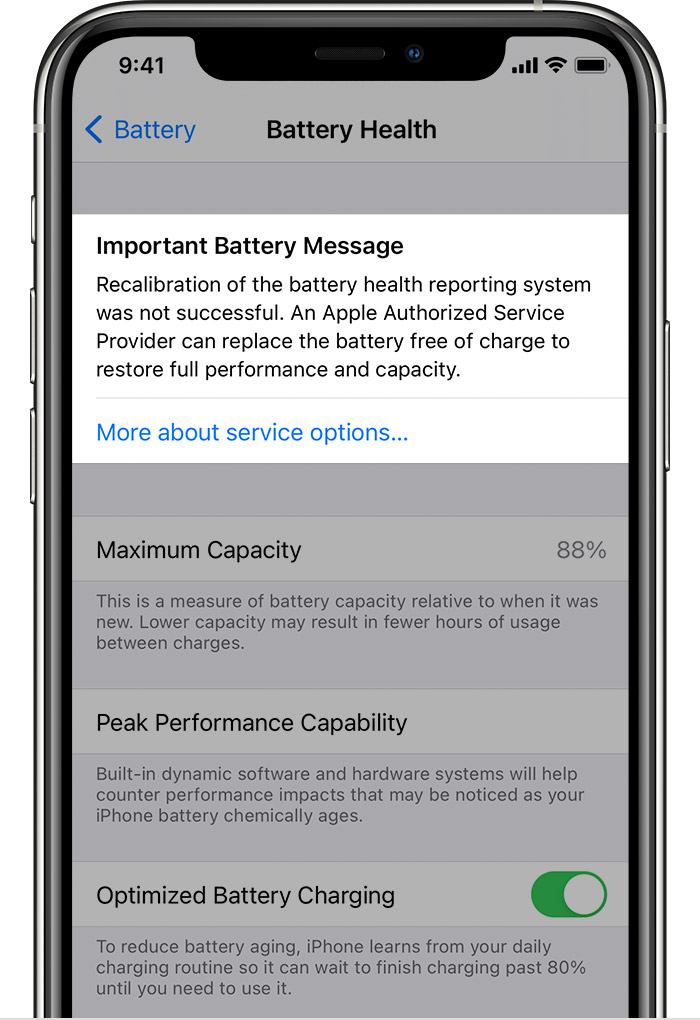
Recalibration of maximum capacity and peak performance capability happens during regular charge cycles, over a few weeks, during which the displayed maximum capacity percentage will not change, though peak performance capability might be updated. If a previous degraded battery message was displayed, this message will be removed after updating to iOS 14.5.
When recalibration is complete, maximum capacity percentage and peak performance capability are updated and the recalibration message removed. If the recalibration still indicates a degraded battery, a battery service message will appear. Should that message be in error (if recalibration fails, for example), an Apple Authorized Service Provider can replace your battery for free.
These are the messages you may see:
- Your battery health reporting system is recalibrating Maximum Capacity and Peak Performance Capability. This process may take a few weeks. Learn more…
- Your battery health reporting system is recalibrating Maximum Capacity and Peak Performance Capability. This process may take a few weeks. Unable to recommend service at this time. Learn more…
- Recalibration of the battery health reporting system was not successful. An Apple Authorized Service Provider can replace the battery free of charge to restore full performance and capacity. More about service options…
How to tell if your Android phone’s battery needs replacing
However, using
AccuBattery
This app is designed to measure actual battery usage with data derived from the battery charge controller.
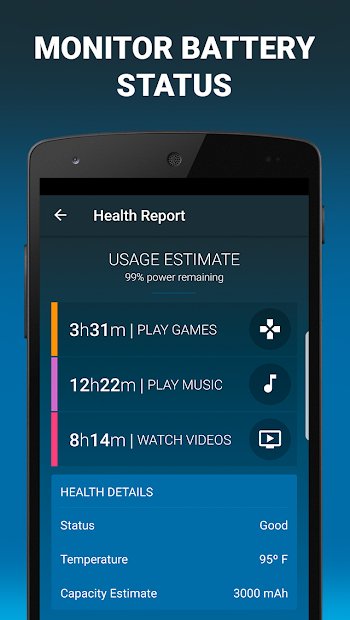
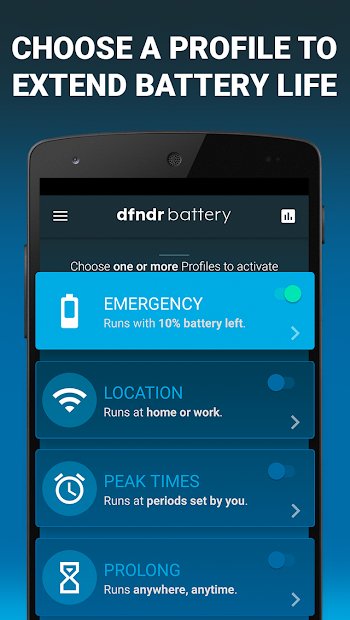
Dfndr Battery
This app gives you an overview of your battery’s overall health and status, with data that includes the percentage of battery power, temperature, and capacity. A battery cooler feature keeps your phone cool by lowering the temperature of your battery and slowing power consumption. It also lets you choose personalized battery saver profiles to customize settings specifically for the way you use your phone.
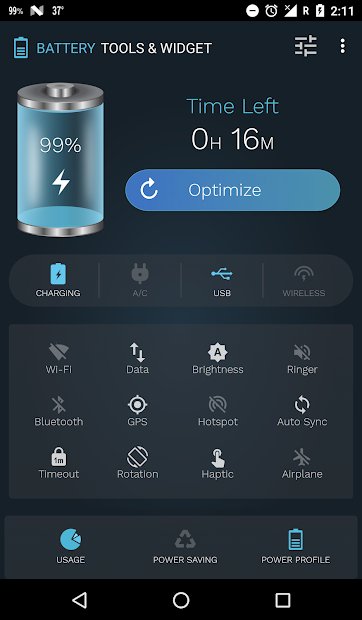
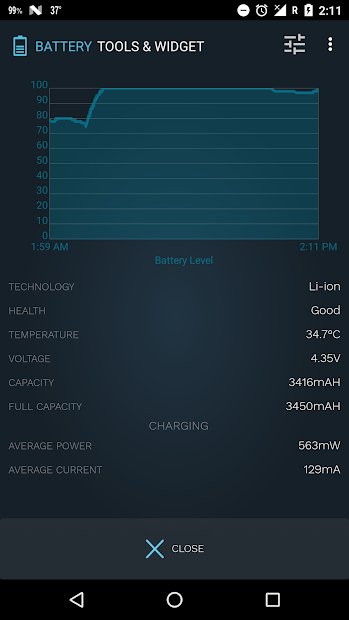
Battery Tools & Widget for Android
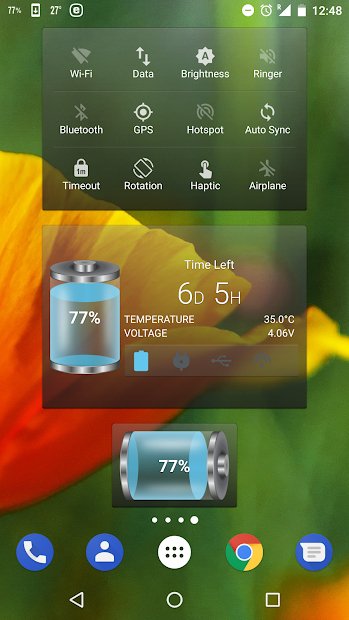
This app manages which features are using power, documents their usage, and maximizes your phone’s battery performance. When you plug your phone in, the app can show you the estimated time left until charging is complete. It also displays battery status, temperature, health, voltage, and a power profile graph. Features include customizable power saving mode, battery saver with optimization, and more.
You’ll need to cease all activity on your phone and take it to a repair shop or genius bar if it is hot to the touch or starts to swell in the middle. It’s better safe than sorry, and you don’t want to deal with something like the dangerous Samsung Note 7 exploding battery issue, in which device batteries overheated and exploded.
Bottom line
If you have a brand new
Editors' Recommendations
- Best iPhone 15 deals: How to get Apple’s latest iPhone for free
- iPhone not holding charge? How to replace an iPhone battery
- The 10 best apps for a second phone number in 2024
- What the green and orange dots on your iPhone really mean
- The best Kindle for reading outside, at night, and more