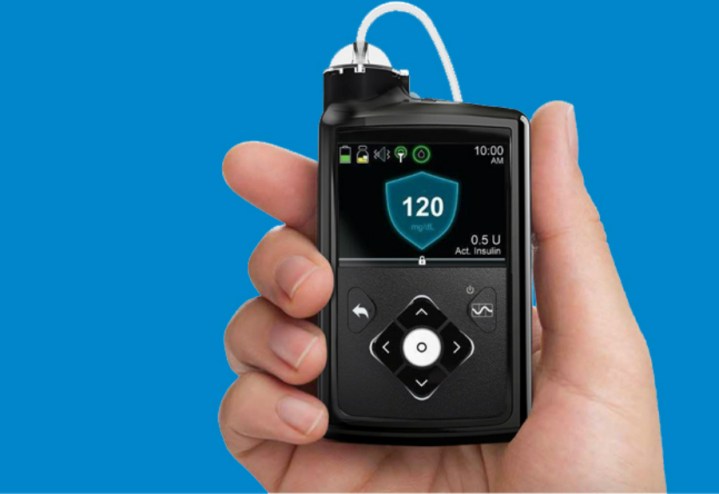
The Medtronic MiniMed 670G system is a wearable device that combines continuous blood-glucose monitoring — blood-sugar testing — with an insulin pump. The algorithm used to adjust insulin dosing, called SmartGuard HCL, was the last step in Medtronic’s phased development of the closed-loop system.
According to Richard M. Bergenstal, M.D., principal investigator of the study and executive director of the Park Nicollet International Diabetes Center in Minneapolis, “The data from the pivotal trial were compelling and I am confident that this therapy will be well-received by both the clinical and patient community.”
The JDRF is enthusiastic as well. “This significant milestone represents an important step forward in the management of Type 1 diabetes and will improve the quality of life for those living with this chronic disease,” said Derek Rapp, JDRF president and CEO. “We are very encouraged by the speed in which this groundbreaking technology was approved by the FDA, and we are proud of the role JDRF played in achieving this exciting breakthrough. Medtronic and JDRF are committed to ensuring appropriate patient access to this therapy.”
The MiniMed 670G system includes Medtronic’s Guardian Sensor glucose monitor, which has a seven-day battery life. In addition to monitoring glucose levels, the Guardian also self-monitors the sensor — if patients rely on the sensor, any breakdown could be fatal, so self-diagnostics are vital. This is the first and so far only sensor approved by the FDA for closed-loop systems. The SmartGuard HCL algorithm has the capability to learn the patient’s insulin needs to be sure glucose levels stay within the optimal target range. The only actions required by patients are to input mealtime carbohydrate intake and periodically calibrate the sensor. No more multiple fingersticks, measurements, and self-administered insulin shots.
Commercial release and availability of the MiniMed 670G are expected in the spring of 2017 and regulatory approval in other countries is expected that summer.



